Mycoplasma PCR Detection & Elimination Bundle
| Cat. No. | G238-G398 |
| Name | Mycoplasma PCR Detection & Elimination Bundle |
| Description |
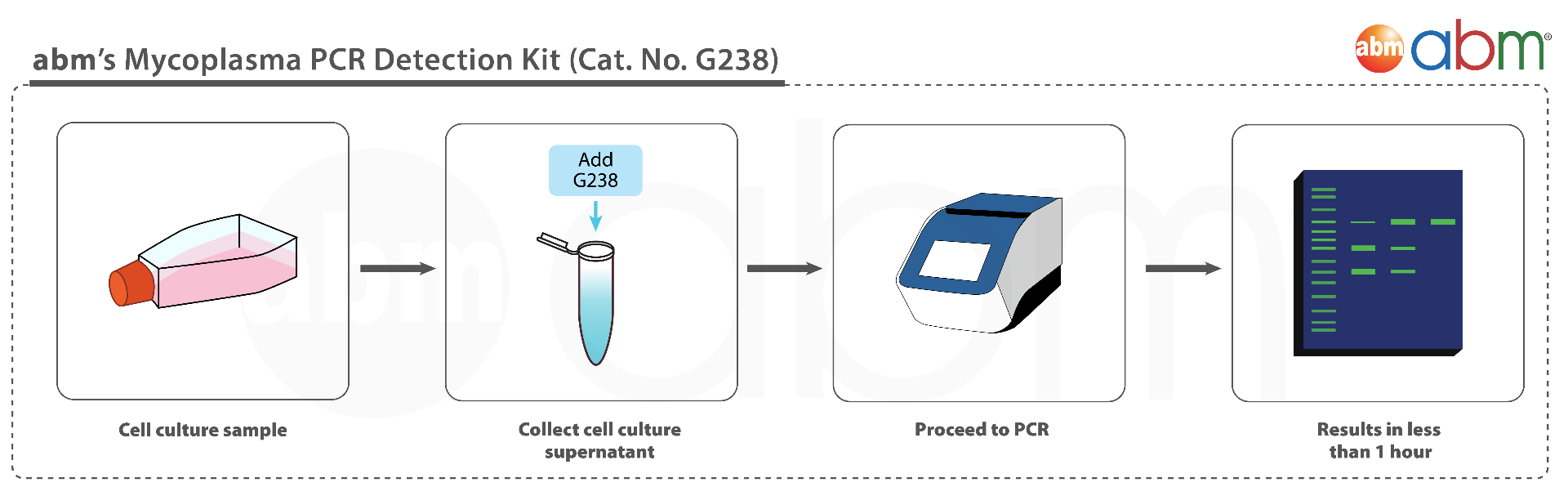
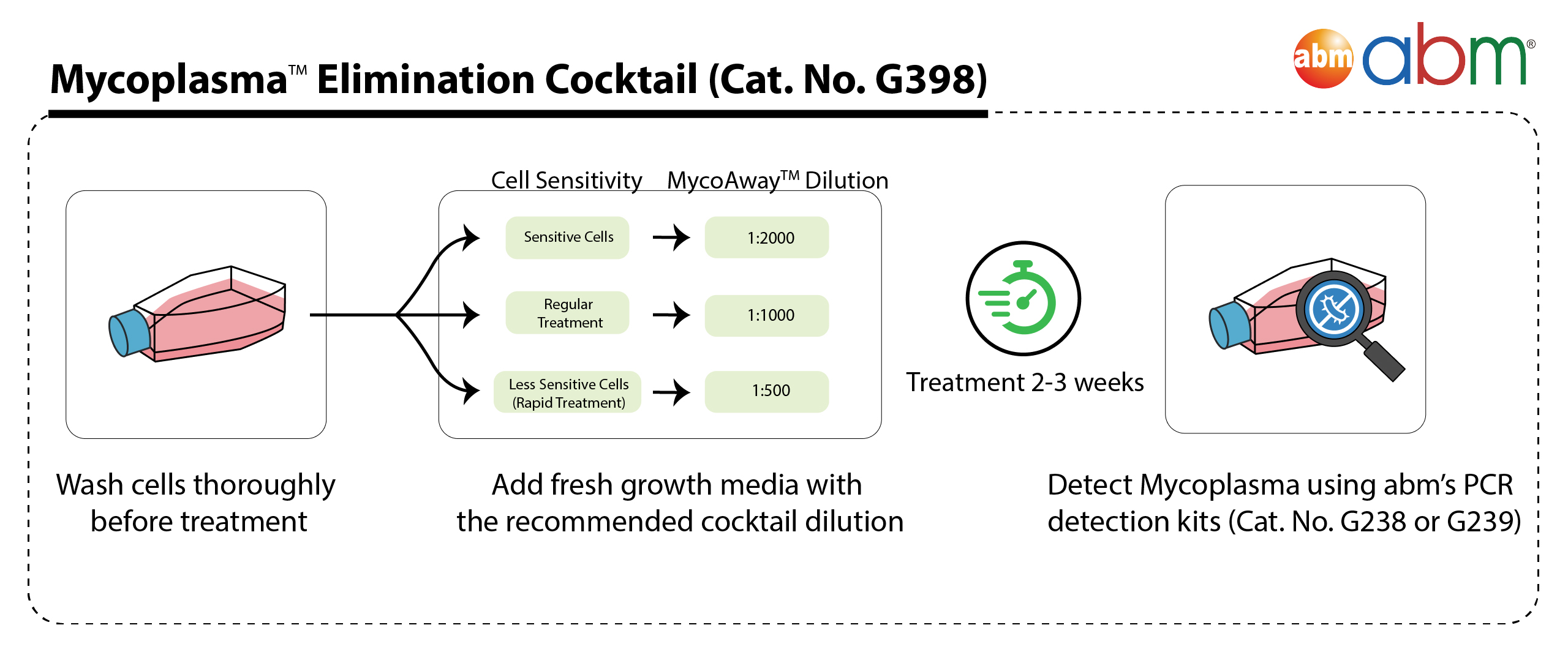
Enjoy superior performance at lower costs! abm's Mycoplasma PCR Detection & Elimination Bundle combines the power of our bestselling kit Mycoplasma PCR Detection Kit (Cat. No. G238) and MycoAway™ Mycoplasma Elimination Cocktail (1000X) (Cat. No. G398). The Mycoplasma PCR Detection Kit offers highly specific and sensitive detection of 200+ strains of Mycoplasma in less than 2 hours. This bestselling kit is designed to minimize false positives, ensuring quick and reliable routine screening of cell cultures. A recent official publication by a national public health agency found that our kit out-performs Sigma and Lonza's kits while being 6X less expensive than ATCC's kit (Russell et al., 2020). Furthermore, the MycoAway™ Mycoplasma Elimination Cocktail (1000X) is a specially formulated agent to effectively clear cells from Mycoplasma contamination in both adherent and suspension cells. Our potent formulation is non-toxic and eliminates 70+ species of Mycoplasma in as little as 2-3 weeks (4-6 cell passages)! The combination of these two kits ensures complete protection of your cell lines! |
| Unit | Bundle |
| Material Citation | If use of this material results in a scientific publication, please cite the material in the following manner: Applied Biological Materials Inc, Cat. No. G238-G398 |
| What is the basis of the positive control in the mycoplasma kit? | |
|
It is DNA fragments of Mycoplasma gDNA diluted in medium.
|
| There is a ~200-250bp band in one of my samples, does this mean it is mycoplasma positive? | |
|
In rare cases, there may be a weak non-specific binding to a region of human gDNA due to the degenerate nature of the primers that can amplify a weak 250bp band in human cell samples. Your sample can be considered as negative for mycoplasma, as mycoplasma amplicons will be 370-550bp in size. You can try to increase the annealing temperature of the PCR program to 60°C to reduce this non-specific amplification.
|
| I did not see amplification of the positive control. What can I try? | |
|
This could occur in cases where the PCR machine does not ramp temperature fast enough. We suggest trying touchdown PCR in these situations.
|
| Can collected supernatant be stored at 4 degrees Celcius until we are ready to run the test? | |
|
Yes, the supernatant can be stored at 4°C for a few weeks.
|
| When collecting supernatant, there is a risk of pipetting cell debris. Can cell debris cause false positive bands? Is centrifugation required? | |
|
Typically centrifugation is not required, however if your sample looks particularly cloudy, you can centrifuge it before use.
|
| After running the PCR program, can the PCR reactions be left in the PCR machine overnight at 12 or 4 degrees Celsius? | |
|
Yes it can be left in the PCR overnight at 12°C or 4°C.
|
| Should PCR components be aliquoted to minimize multiple freeze/thaw cycles? | |
|
For best results, PCR components should be aliquoted if you are frequently testing small quantites of samples. E.g. If you are only testing 1-5 samples each time, aliquoting is recommended to reduce the number of freeze/thaw cycles.
|
| Will the serum in my samples inhibit the PCR reaction? | |
|
Serum will not have any inhibitory effects on PCR. However, if your media contains >20% serum and if it is too viscous, you can dilute with either basal media or Nuclease-free water by tenfold prior to mycoplasma detection.
|
| Can I use this kit for detecting Mycoplasma in various cell culture supernatants? | |
|
Yes, both Mycoplasma PCR Detection Kit (Cat. No. G238) and Mycoplasma Pro Detection Kit (Cat. No. G239) were tested in common laboratory media and serums.
|
| Is there a specific protocol I need to follow when using the kit, or is it straightforward? | |
|
We recommend utilizing the protocol listed in our datasheet since it was specifically developed for our Mycoplasma PCR Detection Kit (Cat. No. G238). If you encounter any challenge detecting Mycoplasma species, consider treating the samples with heat for 5 minutes at 95°C, cooling them down at 4°C, and adding to the reaction mixture.
|
| What are the specific advantages of using your kit compared to other commercially available Mycoplasma detection kits? | |
|
abm’s Mycoplasma PCR Detection Kit (Cat. No. G238) is rapid and comprehensive: more than 200 species will be detected in less than 2 hours. Furthermore, the product is easy-to-use with only 4 components present. Lastly, it is more affordable than other major competitors.
|
| Are there any limitations to the types of Mycoplasma strains that can be detected with this kit? | |
|
Our Mycoplasma PCR Detection Kit (Cat. No. G238) covers more than 200 species, while the Mycoplasma Pro PCR Detection Kit (Cat. No. G239) identifies additional Mycoplasma and Acholeplasma strains such as A. laidlawii. Please refer to our Mycoplasma Strains Guide listed under the Documents section in order to determine which detection kit satisfies your experimental needs.
|
| Can you provide more details on the gel loading dye in the proprietary BlasTaq MasterMix formulation and how it facilitates gel electrophoresis? | |
|
The MasterMix employs Bromophenol Blue sodium salt, a common dye in gel electrophoresis. As the compound is negatively charged, it migrates alongside the DNA making it possible to visualize the PCR product without the need to stain the gel.
|
| Can I expect consistent results over multiple uses, and how many tests can be conducted with a single kit? Can the collected supernatant be stored? If yes, what are the conditions for short-term and long-term storage? | |
|
The results will be consistent over multiple uses. The collected supernatant may be stored in case not all of the cell samples are ready at the same time. We recommend 4°C for short-term storage for up to 1 week and -80°C for long-term storage. Nevertheless, fresh cell culture samples will still provide more conclusive answers.
The kit is sufficient for 100 reactions. |
| Gel electrophoresis shows non-specific amplifications. Any suggestions on how to prevent this? | |
|
Non-specific amplifications can be a result of DNA contamination, excessive templates, and/or wrong annealing temperature. This can be prevented by touchdown PCR.
Touchdown PCR: Lid 105°C Volume 25 μl
1) 95°C for 3min 2) 95°C for 15s 3) 65°C for 15s
4) 72°C for 15s
5) Go to Step 2, x3
6) 95°C for 15s 7) 60°C for 15s
8) 72°C for 15s
9) Go to Step 6, x3
10) 95°C for 15s 11) 55°C for 15s
12) 72°C for 15s
13) Go to Step 10, x25
14) 72°C for 1min 15) 4°C hold
|
| What is the limit of detection of this kit? | |
|
Our kit is able to detect as low as 10 copies of Mycoplasma/sample.
|
| Our cells are grown in suspension and would not achieve 80% confluence, is there a target cell density (cells/ml) we should achieve to ensure we detect the Mycoplasma? | |
|
Mycoplasma testing is more dependent on how long the cells have been in culture. Seeding density does not play a major role when using the detection kit. We recommend cells be kept in culture for 2-3 days without changing the media so as to detect Mycoplasma in culture supernatant.
|
| Will DMSO affect my PCR run? | |
|
As long as the DMSO is diluted and the DMSO content is less than 0.5% in the PCR reaction it should not cause a problem. Too much DMSO would alter the Tm of the primers and thus affect amplification.
|
| My culture media contains antibiotics, will that be an issue? | |
|
It is fine to test supernatant containing antibiotics. Please ensure that the culture has been in incubation for at least 48-72 hours prior to sample collection.
|
-
Bahreyni, A., Liu, H., Mohamud, Y., Xue, Y. C., Zhang, J., & Luo, H. (2022). A new miRNA-Modified coxsackievirus B3 inhibits triple negative breast cancer growth with improved safety profile in immunocompetent mice. Cancer Letters, 548, 215849. https://doi.org/10.1016/j.canlet.2022.215849
Conrad, S. J., Silva, R. F., Hearn, C. J., Climans, M., & Dunn, J. R. (2018). Attenuation of Marek's disease virus by codon pair deoptimization of a core gene. Virology, 516, 219-226. https://doi.org/10.1016/j.virol.2018.01.020
Dogra, S., Elayapillai, S. P., Qu, D., Pitts, K., Filatenkov, A., Houchen, C. W., ... & Hannafon, B. N. (2023). Targeting doublecortin-like kinase 1 reveals a novel strategy to circumvent chemoresistance and metastasis in ovarian cancer. Cancer Letters, 578, 216437. https://doi.org/10.1016/j.canlet.2023.216437
Du, L., Liu, W., Aldana-Masangkay, G. et al. SUMOylation inhibition enhances dexamethasone sensitivity in multiple myeloma. J Exp Clin Cancer Res 41, 8 (2022). https://doi.org/10.1186/s13046-021-02226-9
Favaretto, G., Rossi, M.N., Cuollo, L. et al. Neutrophil-activating secretome characterizes palbociclib-induced senescence of breast cancer cells. Cancer Immunol Immunother 73, 113 (2024). https://doi.org/10.1007/s00262-024-03695-5
Jia, B., Yin, X., Wang, Y., Qian, J., He, Y., Yang, C., … Meng, X. (2020). CircRNA-PTN Sponges miR-326 to Promote Proliferation in Hepatocellular Carcinoma. OncoTargets and Therapy, 13, 4893–4903. https://doi.org/10.2147/OTT.S251300
Kalejaiye, T. D., Bhattacharya, R., Burt, M. A., Travieso, T., Okafor, A. E., Mou, X., ... & Musah, S. (2022). SARS-CoV-2 employ BSG/CD147 and ACE2 receptors to directly infect human induced pluripotent stem cell-derived kidney podocytes. Frontiers in Cell and Developmental Biology, 10, 855340. https://doi.org/10.3389/fcell.2022.855340
Kurden-Pekmezci, A., Cakiroglu, E., Eris, S., Mazi, F. A., Coskun-Deniz, O. S., Dalgic, E., ... & Senturk, S. (2023). MALT1 paracaspase is overexpressed in hepatocellular carcinoma and promotes cancer cell survival and growth. Life Sciences, 323, 121690. https://doi.org/10.1016/j.lfs.2023.121690
Lee, M. H., Ratanachan, D., Wang, Z., Hack, J., Adbulrahman, L., Shamlin, N. P., ... & Schaue, D. (2023). Adaptation of the tumor antigen presentation machinery to ionizing radiation. The Journal of Immunology, 211(4), 693-705. https://doi.org/10.4049/jimmunol.2100793
Li, F., Kitajima, S., Kohno, S., Yoshida, A., Tange, S., Sasaki, S., ... & Takahashi, C. (2019). Retinoblastoma inactivation induces a protumoral microenvironment via enhanced CCL2 secretion. Cancer Research, 79(15), 3903-3915. https://doi.org/10.1158/0008-5472.CAN-18-3604
Lorusso, B., Cerasoli, G., Falco, A., Frati, C., Graiani, G., Madeddu, D., ... & Lagrasta, C. (2022). Β-blockers activate autophagy on infantile hemangioma-derived endothelial cells in vitro. Vascular pharmacology, 146, 107110. https://doi.org/10.1016/j.vph.2022.107110
Miranda, L., Mandrich, L., Massa, S., Nutile, T., Crovella, C., De Rosa, I., ... & Caputo, E. (2025). Breast Cancer Tissues and Organoids BioBank: Constitution, Research Activities and Samples Access. Organoids, 4(1), 5. https://doi.org/10.3390/organoids4010005
Nasser, A. M., Melamed, L., Wetzel, E. A., Chang, J. C. C., Nagashima, H., Kitagawa, Y., ... & Miller, J. J. (2024). CDKN2A/B homozygous deletion sensitizes IDH-mutant glioma to CDK4/6 inhibition. Clinical Cancer Research, 30(14), 2996-3005. https://doi.org/10.1158/1078-0432.CCR-24-0562
Pelullo, M., Nardozza, F., Zema, S., Quaranta, R., Nicoletti, C., Besharat, Z. M., ... & Bellavia, D. (2019). Kras/ADAM17-dependent Jag1-ICD reverse signaling sustains colorectal cancer progression and chemoresistance. Cancer research, 79(21), 5575-5586. https://doi.org/10.1158/0008-5472.CAN-19-0145
Pelullo, M., Zema, S., De Carolis, M., Cialfi, S., Giuli, M. V., Palermo, R., ... & Bellavia, D. (2022). 5FU/Oxaliplatin-induced jagged1 cleavage counteracts apoptosis induction in colorectal cancer: A novel mechanism of intrinsic drug resistance. Frontiers in Oncology, 12, 918763. https://doi.org/10.3389/fonc.2022.918763
Roye, Y., Bhattacharya, R., Mou, X., Zhou, Y., Burt, M. A., & Musah, S. (2021). A personalized glomerulus chip engineered from stem cell-derived epithelium and vascular endothelium. Micromachines, 12(8), 967. https://doi.org/10.3390/mi12080967
Zhong, N., Li, D., Wang, B., Kovalchuk, O., & Kovalchuk, I. (2023). Cannabinol inhibits cell growth and triggers cell cycle arrest and apoptosis in cancer cells. Biocatalysis and Agricultural Biotechnology, 48, 102627. https://doi.org/10.1016/j.bcab.2023.102627
Zhou, Q., Pichlmeier, S., Denz, A. M., Schreiner, N., Straub, T., Benitz, S., ... & Regel, I. (2024). Altered histone acetylation patterns in pancreatic cancer cell lines induce subtype‑specific transcriptomic and phenotypical changes. International journal of oncology, 64(3), 1-12. https://doi.org/10.3892/ijo.2024.5614
Zhu, Q., Zhang, X., Lu, F. et al. RUNX1-BMP2 promotes vasculogenic mimicry in laryngeal squamous cell carcinoma via activation of the PI3K-AKT signaling pathway. Cell Commun Signal 22, 227 (2024). https://doi.org/10.1186/s12964-024-01605-x
-
Eyme, K. M., Carvalho, L., & Badr, C. E. (2021). Intranasal delivery of experimental compounds in orthotopic brain tumor mouse models. STAR protocols, 2(1), 100290. https://doi.org/10.1016/j.xpro.2020.100290
Eyme, K. M., Sammarco, A., & Badr, C. E. (2022). Orthotopic brain tumor models derived from glioblastoma stem-like cells. In Methods in Cell Biology (Vol. 170, pp. 1-19). Academic Press. https://doi.org/10.1016/bs.mcb.2022.02.003