TEV Protease
|
Cat. No.
|
E027
Print
|
|
Name
|
TEV Protease
|
|
Unit
|
100 μl
|
|
Category
|
Molecular Biology Enzymes and Kits
|
|
Description
|
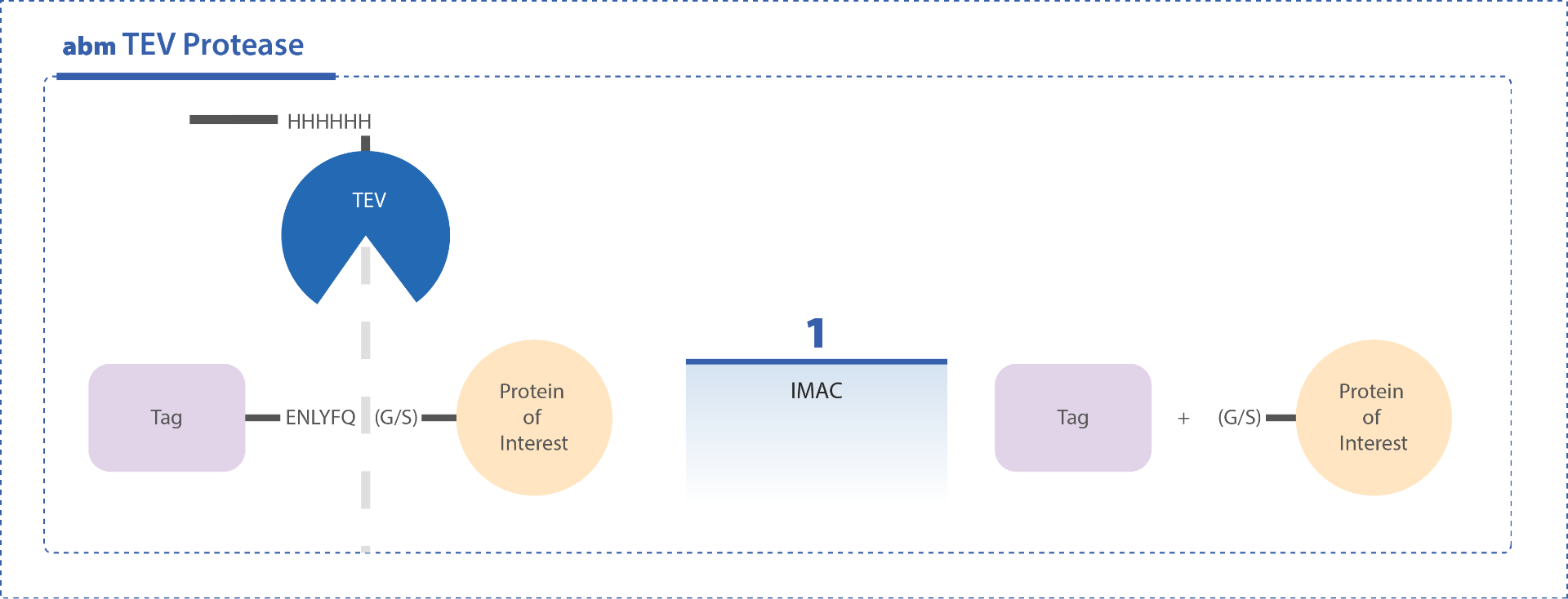
abm’s TEV Protease is an enhanced version of the site-specific protease derived from Tobacco Etch Virus (TEV). This optimized enzyme offers superior activity, stability, and site-specificity compared to the native form. TEV Protease cleaves fusion proteins with high precision at the Gln-Gly or Gln-Ser bond within the seven-amino acid recognition sequence Glu-Asn-Leu-Tyr-Phe-Gln-Gly/Ser (ENLYFQ(G/S)).
The enzyme is highly effective across a broad range of temperatures (4–30°C, with an optimum at 30°C) and pH values (5.5–9.0), enabling flexible experimental conditions. Under optimal conditions, up to 99% cleavage can be achieved in just 1–2 hours. Additionally, the inclusion of a 6X-His tag at the N-terminus allows for convenient removal of TEV Protease post-cleavage using Ni-IDA Agarose Beads (Cat. No. G250) via affinity chromatography.
| Product Component |
Quantity |
| TEV Protease (10 U/µl) |
100 µl |
| 20X TEV Protease Reaction Buffer |
1.0 ml |
| 100 mM DTT |
500 µl |
|
|
Application
|
- Cleavage of tags from recombinant fusion proteins containing a TEV recognition site
- One step affinity removal of His-tagged TEV after cleavage
|
|
Concentration
|
10U/ul
|
|
Material Citation
|
If use of this material results in a scientific publication, please cite the material in the following manner: Applied Biological Materials Inc, Cat. No. E027
|
|
|
What is an Enzyme Unit defined as?
|
|
One unit is defined as the amount of TEV Protease that is required to cleave >90% of 3 µg of control substrate in a 30 µl reaction for 1 hour at 30°C in 1X TEV Protease Reaction Buffer supplemented with 1 mM DTT.
|
-
Issaro, N., Kongkaew, A., Jittmittraphap, A., Leaungwutiwong, P., Nimlamool, W., & Takuathung, M. N. (2023). Expression of polyprotein and 3D polymerase protein in Sf9 cells and immunogenicity against enterovirus A71B5 (Thailand strain). Journal of Applied Pharmaceutical Science, 13(9), 027-036. https://dx.doi.org/10.7324/JAPS.2023.93192
Liu, H., Yamaguchi, H., Kikkawa, M., & Shima, T. (2024). Heterogeneous local structures of the microtubule lattice revealed by cryo-ET and non-averaging analysis. bioRxiv, 2024-04. https://doi.org/10.1101/2024.04.30.591984
Mori, S., Nagae, M., & Yamasaki, S. (2024). Crystal structure of the complex of CLEC12A and an antibody that interferes with binding of diverse ligands. International Immunology, 36(6), 279-290. https://doi.org/10.1093/intimm/dxae006
Ogasawara, S., & Yamada, A. (2022). RNA editing with viral RNA-dependent RNA polymerase. ACS Synthetic Biology, 11(1), 46-52. https://doi.org/10.1021/acssynbio.1c00332
Takagi, M., Nagatani, A., Kawano, K., Hata, A., Yokoyama, A., Hayashida, K., ... & Matsuzaki, K. (2024). Stable and Minimum Size Solubilization of Membrane Proteins with Cocktails of Phospholipid Analogues. ACS Applied Materials & Interfaces, 16(46), 63358-63367. https://doi.org/10.1021/acsami.4c15697
Takahasi, K., Onomoto, K., Horiuchi, M., Kato, H., Fujita, T., & Yoneyama, M. (2019). Identification of a new autoinhibitory domain of interferon-beta promoter stimulator-1 (IPS-1) for the tight regulation of oligomerization-driven signal activation. Biochemical and Biophysical Research Communications, 517(4), 662-669. https://doi.org/10.1016/j.bbrc.2019.07.099
Tobita, Y., Hirano, K., Miura, D., Hatano, Y., Tsugawa, W., Ikebukuro, K., ... & Asano, R. (2025). A Versatile Method to Create Antibody/Split‐Enzyme Complexes and Its Application to a Rapid, Homogeneous, and Universal Electrochemical Immunosensing System. Advanced Sensor Research, 4(1), 2400112. https://doi.org/10.1002/adsr.202400112
This product has no review yet.
Controls and Related Product:
{"id":755,"cat_no":"E027","sku_in_m2":"","old_cat_no":"E027","name":"TEV Protease","category_id":186,"category_name":"Molecular Biology Enzymes and Kits","frontend_category_id":6,"frontend_category_ids":"1,6","gene_id":0,"gene_name":"","gene_full_name":"","alias":"","unit_quantity":"100 \u03bcl","accession_number":"","species_id":0,"species":"","raised_in_id":0,"raised_in":"","cell_type_id":0,"cell_type":"","tissue_id":0,"tissue":"","price":"49.0000","price_before_discount":"0.0000","keywords":"0","invisible_search_keywords":"","filter_group_id":34,"filter_group_name":"Molecular Biology","filter_id":41,"filter_name":"Kits \u0026 Other Enzymes","search_filter_ids":"32,34","search_filter_keys":"32-34","temp_field":null,"customization":0,"filter_table_id":18084831,"filter_table_name":"","related_group_id":17,"url":"","operator":"","points_rule":7,"status":1,"need_remove":0,"document_sort":"","price_old":"45.0000","price2024":"47.5000","created_at":null,"updated_at":"2025-03-26 17:31:22","Confirmed":"Tony Confirmed","geo_price_before_discount":"0.00","geo_titer7_price_before_discount":"149.00","cate_url":"Molecular-Biology-Enzymes-and-Kits.html","frontendBreadcrumbs":[{"id":1,"pid":86,"name":"General Materials","page_id":27,"page_type":"App\\Models\\Category","href":null,"sort":1,"show_in_nav":1,"show_in_homepage":1,"has_children":1,"status":1,"related_table":null,"created_at":null,"updated_at":null,"url_key":"general-materials.html","url":"general-materials.html"},{"id":6,"pid":1,"name":"Enzymes \u0026 Kits","page_id":186,"page_type":"App\\Models\\Category","href":null,"sort":6,"show_in_nav":1,"show_in_homepage":1,"has_children":1,"status":1,"related_table":null,"created_at":null,"updated_at":null,"url_key":"Molecular-Biology-Enzymes-and-Kits.html","url":"Molecular-Biology-Enzymes-and-Kits.html"}],"price_flag":0,"builderURL":"","builderViewerURL":"","mapURL":"","viewerURL":"","builderType":"","productType":"App\\Models\\CatalogBaseMolecular","geo_price":"49.00","geo_price_symbol":"$49.00","category":{"id":186,"categories":"Molecular Biology Enzymes and Kits","pid":27,"url_key":"Molecular-Biology-Enzymes-and-Kits.html","css":"\/assets\/css\/abm.search.css,\/assets\/css\/abm.other.css,\/assets\/css\/abm.categorys.css, \/assets\/css\/ryan.temp.css","wid":0,"breadcrumbs":"[{\u0022url\u0022:\u0022https:\/\/www.abmgood.com\u0022,\u0022title\u0022:\u0022Home\u0022},{\u0022url\u0022:\u0022https:\/\/www.abmgood.com\/general-materials.html\u0022,\u0022title\u0022:\u0022General Materials \u0026amp; Others\u0022},{\u0022url\u0022:\u0022https:\/\/www.abmgood.com\/Molecular-Biology-Enzymes-and-Kits.html\u0022,\u0022title\u0022:\u0022Molecular Biology Enzymes and Kits\u0022}]","breadcrumbsIdJson":"\u003Cscript type=\u0022application\/ld+json\u0022\u003E{\u0022@context\u0022:\u0022https:\/\/schema.org\u0022,\u0022@type\u0022:\u0022BreadcrumbList\u0022,\u0022itemListElement\u0022:[{\u0022@type\u0022:\u0022ListItem\u0022,\u0022position\u0022:1,\u0022name\u0022:\u0022Home\u0022,\u0022item\u0022:\u0022https:\/\/www.abmgood.com\u0022},{\u0022@type\u0022:\u0022ListItem\u0022,\u0022position\u0022:2,\u0022name\u0022:\u0022General Materials \u0026amp; Others\u0022,\u0022item\u0022:\u0022https:\/\/www.abmgood.com\/general-materials.html\u0022},{\u0022@type\u0022:\u0022ListItem\u0022,\u0022position\u0022:3,\u0022name\u0022:\u0022Molecular Biology Enzymes and Kits\u0022}]}\u003C\/script\u003E","breadcrumbsHtml":"\u003Cul class=\u0022abm-breadcrumb\u0022\u003E\u003Cli\u003E\u003Ca href=\u0022https:\/\/www.abmgood.com\u0022\u003EHome\u003C\/a\u003E\u003C\/li\u003E\u003Cli\u003E\u003Ca href=\u0022https:\/\/www.abmgood.com\/general-materials.html\u0022\u003EGeneral Materials \u0026amp; Others\u003C\/a\u003E\u003C\/li\u003E\u003Cli class=\u0022active\u0022\u003EMolecular Biology Enzymes and Kits\u003C\/li\u003E\u003C\/ul\u003E","description":"\u003Ch2 class=\u0022abm-categories-title-h2\u0022\u003EMolecular Biology Enzymes and Kits\u003C\/h2\u003E\n\u003Cul class=\u0022abm-page-category-nav-list\u0022 style=\u0022margin-top: -20px;\u0022\u003E\n\u003Cli class=\u0022abm-page-category-nav-item\u0022\u003E\u003Ca href=\u0022#Cloning-Applications\u0022 class=\u0022abm-page-category-nav-link abm-page-category-target-link\u0022\u003E Cloning Applications\u003C\/a\u003E\u003C\/li\u003E\n\u003Cli class=\u0022abm-page-category-nav-item\u0022\u003E\u003Ca href=\u0022#DNA-RNA-Applications\u0022 class=\u0022abm-page-category-nav-link abm-page-category-target-link\u0022\u003E DNA \u0026amp; RNA Applications\u003C\/a\u003E\u003C\/li\u003E\n\u003Cli class=\u0022abm-page-category-nav-item\u0022\u003E\u003Ca href=\u0022#in-vitro-Kits-Proteases\u0022 class=\u0022abm-page-category-nav-link abm-page-category-target-link\u0022\u003EProteases\u003C\/a\u003E\u003C\/li\u003E\n\u003Cli class=\u0022abm-page-category-nav-item\u0022\u003E\u003Ca href=\u0022\/rnase-r-e049.html\u0022 class=\u0022abm-page-category-nav-link abm-page-category-target-link\u0022\u003E RNase R\u003C\/a\u003E\u003C\/li\u003E\n\u003Cli class=\u0022abm-page-category-nav-item\u0022\u003E\u003Ca href=\u0022\/cas9-proteins.html\u0022 class=\u0022abm-page-category-nav-link abm-page-category-target-link\u0022\u003ECas Proteins \u0026amp; CRISPR Screening\u003C\/a\u003E\u003C\/li\u003E\n\u003C\/ul\u003E\n\u003Cdiv class=\u0022abm-container-fluid\u0022\u003E\n\u003Cdiv class=\u0022row\u0022\u003E\n\u003Cdiv class=\u0022col-md-12\u0022 style=\u0022text-align: left; padding-bottom: 3em;\u0022\u003E\n\u003Cp\u003E\u003Cstrong\u003Eabm\u003C\/strong\u003E offers a variety of high quality individual enzymes and kits that enable the preparation of DNA and RNA for subsequent downstream applications such as assay development, PCR, restriction digest, cloning and sequencing. Simplify your experiments with our trusted products for consistent, high-performance results.\u003C\/p\u003E\n\u003Cdiv class=\u0022table-responsive\u0022\u003E\n\u003Ctable class=\u0022bootstrap-table bootstrap-table-hover abm-service-table abm-perfect-table1\u0022 style=\u0022--first-child-white-space: nowrap;\u0022\u003E\n\u003Cthead\u003E\n\u003Ctr\u003E\n\u003Cth\u003EProduct Name\u003C\/th\u003E\n\u003Cth\u003ECat. No.\u003C\/th\u003E\n\u003Cth\u003ESize\u003C\/th\u003E\n\u003Cth\u003EPrice\u003C\/th\u003E\n\u003C\/tr\u003E\n\u003C\/thead\u003E\n\u003Ctbody\u003E\n\u003Ctr style=\u0022background-color: #f8f9fa;\u0022 align=\u0022left\u0022 class=\u0022abm-page-menu only-hide\u0022 id=\u0022Cloning-Applications\u0022\u003E\n\u003Ctd colspan=\u00224\u0022 style=\u0022text-align: left;\u0022\u003E\u003Cspan style=\u0022color: #000;\u0022\u003E\u003Cstrong\u003ECloning Applications\u003C\/strong\u003E\u003C\/span\u003E\u003C\/td\u003E\n\u003C\/tr\u003E\n\u003Ctr\u003E\n\u003Ctd style=\u0022text-align: left;\u0022\u003E\u003Ca class=\u0022orange-link\u0022 href=\u0022\/pro-ligation-free-cloning-kit-e086.html\u0022 rel=\u0022noopener\u0022\u003EPro Ligation-Free Cloning Kit (20 reactions)\u003C\/a\u003E\u003C\/td\u003E\n\u003Ctd style=\u0022text-align: left;\u0022\u003E\u003Ca class=\u0022orange-link\u0022 href=\u0022\/pro-ligation-free-cloning-kit-e086.html\u0022 rel=\u0022noopener\u0022\u003EE086\u003C\/a\u003E\u003C\/td\u003E\n\u003Ctd style=\u0022text-align: left;\u0022\u003E20 Reactions\u003C\/td\u003E\n\u003Ctd class=\u0022product-price\u0022 data-product=\u0022E086\u0022 data-type=\u0022molecular\u0022\u003E$150.00\u003C\/td\u003E\n\u003C\/tr\u003E\n\u003Ctr\u003E\n\u003Ctd style=\u0022text-align: left;\u0022\u003E\u003Ca class=\u0022orange-link\u0022 href=\u0022\/pro-ligation-free-cloning-kit-e087.html\u0022 rel=\u0022noopener\u0022\u003EPro Ligation-Free Cloning Kit (50 reactions)\u003C\/a\u003E\u003C\/td\u003E\n\u003Ctd style=\u0022text-align: left;\u0022\u003E\u003Ca class=\u0022orange-link\u0022 href=\u0022\/pro-ligation-free-cloning-kit-e087.html\u0022 rel=\u0022noopener\u0022\u003EE087\u003C\/a\u003E\u003C\/td\u003E\n\u003Ctd style=\u0022text-align: left;\u0022\u003E50 Reactions\u003C\/td\u003E\n\u003Ctd class=\u0022product-price\u0022 data-product=\u0022E087\u0022 data-type=\u0022molecular\u0022\u003E$250.00\u003C\/td\u003E\n\u003C\/tr\u003E\n\u003Ctr\u003E\n\u003Ctd style=\u0022text-align: left;\u0022\u003E\u003Ca class=\u0022orange-link\u0022 href=\u0022\/proclone-competent-cells-3.html\u0022 rel=\u0022noopener\u0022\u003EProClone\u0026trade; Competent Cells\u003C\/a\u003E\u003C\/td\u003E\n\u003Ctd style=\u0022text-align: left;\u0022\u003E\u003Ca class=\u0022orange-link\u0022 href=\u0022\/proclone-competent-cells-3.html\u0022 rel=\u0022noopener\u0022\u003EE003\u003C\/a\u003E\u003C\/td\u003E\n\u003Ctd style=\u0022text-align: left;\u0022\u003E1.2 ml (6 x 200 \u0026micro;l)\u003C\/td\u003E\n\u003Ctd class=\u0022product-price\u0022 data-product=\u0022E003\u0022 data-type=\u0022molecular\u0022\u003E$150.00\u003C\/td\u003E\n\u003C\/tr\u003E\n\u003Ctr\u003E\n\u003Ctd style=\u0022text-align: left;\u0022\u003E\u003Ca class=\u0022orange-link\u0022 href=\u0022\/phi29-dna-polymerase-e014.html\u0022 rel=\u0022noopener\u0022\u003EUltra-Pure Phi29 DNA Polymerase\u003C\/a\u003E\u003C\/td\u003E\n\u003Ctd style=\u0022text-align: left;\u0022\u003E\u003Ca class=\u0022orange-link\u0022 href=\u0022\/phi29-dna-polymerase-e014.html\u0022 rel=\u0022noopener\u0022\u003EE014\u003C\/a\u003E\u003C\/td\u003E\n\u003Ctd style=\u0022text-align: left;\u0022\u003E100 \u0026micro;l\u003C\/td\u003E\n\u003Ctd class=\u0022product-price\u0022 data-product=\u0022E014\u0022 data-type=\u0022molecular\u0022\u003E$152.00\u003C\/td\u003E\n\u003C\/tr\u003E\n\u003Ctr style=\u0022background-color: #f8f9fa;\u0022 align=\u0022left\u0022 class=\u0022abm-page-menu only-hide\u0022 id=\u0022DNA-RNA-Applications\u0022\u003E\n\u003Ctd colspan=\u00224\u0022 style=\u0022text-align: left;\u0022\u003E\u003Cspan style=\u0022color: #000;\u0022\u003E\u003Cstrong\u003EDNA \u0026amp; RNA Applications\u003C\/strong\u003E\u003C\/span\u003E\u003C\/td\u003E\n\u003C\/tr\u003E\n\u003Ctr\u003E\n\u003Ctd style=\u0022text-align: left;\u0022\u003E\u003Ca class=\u0022orange-link\u0022 href=\u0022\/rnase-r-e049.html\u0022 rel=\u0022noopener\u0022\u003ERNase R\u003C\/a\u003E\u003C\/td\u003E\n\u003Ctd style=\u0022text-align: left;\u0022\u003E\u003Ca class=\u0022orange-link\u0022 href=\u0022\/rnase-r-e049.html\u0022 rel=\u0022noopener\u0022\u003EE049\u003C\/a\u003E\u003C\/td\u003E\n\u003Ctd style=\u0022text-align: left;\u0022\u003E500 U (50 \u0026mu;l)\u003C\/td\u003E\n\u003Ctd class=\u0022product-price\u0022 data-product=\u0022E049\u0022 data-type=\u0022molecular\u0022\u003E$310.00\u003C\/td\u003E\n\u003C\/tr\u003E\n\u003Ctr\u003E\n\u003Ctd style=\u0022text-align: left;\u0022\u003E\u003Ca class=\u0022orange-link\u0022 href=\u0022\/rnaseoff-ribonuclease-inhibitor.html\u0022 rel=\u0022noopener\u0022\u003ERNaseOFF Ribonuclease Inhibitor\u003C\/a\u003E\u003C\/td\u003E\n\u003Ctd style=\u0022text-align: left;\u0022\u003E\u003Ca class=\u0022orange-link\u0022 href=\u0022\/rnaseoff-ribonuclease-inhibitor.html\u0022 rel=\u0022noopener\u0022\u003EG138\u003C\/a\u003E\u003C\/td\u003E\n\u003Ctd style=\u0022text-align: left;\u0022\u003E4,000 U (100 \u0026micro;l)\u003C\/td\u003E\n\u003Ctd class=\u0022product-price\u0022 data-product=\u0022G138\u0022 data-type=\u0022molecular\u0022\u003E$80.00\u003C\/td\u003E\n\u003C\/tr\u003E\n\u003Ctr\u003E\n\u003Ctd style=\u0022text-align: left;\u0022\u003E\u003Ca class=\u0022orange-link\u0022 href=\u0022\/rnaseoff-ribonuclease-inhibitor-g591.html\u0022 rel=\u0022noopener\u0022\u003ERNaseOFF Ribonuclease Inhibitor\u003C\/a\u003E\u003C\/td\u003E\n\u003Ctd style=\u0022text-align: left;\u0022\u003E\u003Ca class=\u0022orange-link\u0022 href=\u0022\/rnaseoff-ribonuclease-inhibitor-g591.html\u0022 rel=\u0022noopener\u0022\u003EG591\u003C\/a\u003E\u003C\/td\u003E\n\u003Ctd style=\u0022text-align: left;\u0022\u003E100,000 U (2 x 1.25 ml)\u003C\/td\u003E\n\u003Ctd class=\u0022product-price\u0022 data-product=\u0022G591\u0022 data-type=\u0022molecular\u0022\u003E$1400.00\u003C\/td\u003E\n\u003C\/tr\u003E\n\u003Ctr\u003E\n\u003Ctd style=\u0022text-align: left;\u0022\u003E\u003Ca class=\u0022orange-link\u0022 href=\u0022\/rnaser-rnaseoff-bundle.html\u0022 rel=\u0022noopener\u0022\u003ERNase R with RNaseOFF Bundle\u003C\/a\u003E\u003C\/td\u003E\n\u003Ctd style=\u0022text-align: left;\u0022\u003E\u003Ca class=\u0022orange-link\u0022 href=\u0022\/rnaser-rnaseoff-bundle.html\u0022 rel=\u0022noopener\u0022\u003EE049-G138S\u003C\/a\u003E\u003C\/td\u003E\n\u003Ctd style=\u0022text-align: left;\u0022\u003EBundle\u003C\/td\u003E\n\u003Ctd class=\u0022product-price\u0022 data-product=\u0022E049-G138S\u0022 data-type=\u0022molecular\u0022\u003E$349.00\u003C\/td\u003E\n\u003C\/tr\u003E\n\u003Ctr\u003E\n\u003Ctd style=\u0022text-align: left;\u0022\u003E\u003Ca class=\u0022orange-link\u0022 href=\u0022\/rnase-a-250.html\u0022 rel=\u0022noopener\u0022\u003ERNase A\u003C\/a\u003E\u003C\/td\u003E\n\u003Ctd style=\u0022text-align: left;\u0022\u003E\u003Ca class=\u0022orange-link\u0022 href=\u0022\/rnase-a-250.html\u0022 rel=\u0022noopener\u0022\u003EG117\u003C\/a\u003E\u003C\/td\u003E\n\u003Ctd style=\u0022text-align: left;\u0022\u003E25 mg\u003C\/td\u003E\n\u003Ctd class=\u0022product-price\u0022 data-product=\u0022G117\u0022 data-type=\u0022molecular\u0022\u003E$57.50\u003C\/td\u003E\n\u003C\/tr\u003E\n\u003Ctr\u003E\n\u003Ctd style=\u0022text-align: left;\u0022\u003E\u003Ca class=\u0022orange-link\u0022 href=\u0022\/dnase-i-g028.html\u0022 rel=\u0022noopener\u0022\u003EDNase I\u003C\/a\u003E\u003C\/td\u003E\n\u003Ctd style=\u0022text-align: left;\u0022\u003E\u003Ca class=\u0022orange-link\u0022 href=\u0022\/dnase-i-g028.html\u0022 rel=\u0022noopener\u0022\u003EG028\u003C\/a\u003E\u003C\/td\u003E\n\u003Ctd style=\u0022text-align: left;\u0022\u003E2000 U (1.0 ml)\u003C\/td\u003E\n\u003Ctd class=\u0022product-price\u0022 data-product=\u0022G028\u0022 data-type=\u0022molecular\u0022\u003E$89.00\u003C\/td\u003E\n\u003C\/tr\u003E\n\u003Ctr\u003E\n\u003Ctd style=\u0022text-align: left;\u0022\u003E\u003Ca class=\u0022orange-link\u0022 href=\u0022\/dnase-i-rnase-free-e091.html\u0022 rel=\u0022noopener\u0022\u003EDNase I (RNase-Free)\u003C\/a\u003E\u003C\/td\u003E\n\u003Ctd style=\u0022text-align: left;\u0022\u003E\u003Ca class=\u0022orange-link\u0022 href=\u0022\/dnase-i-rnase-free-e091.html\u0022 rel=\u0022noopener\u0022\u003EE091\u003C\/a\u003E\u003C\/td\u003E\n\u003Ctd style=\u0022text-align: left;\u0022\u003E200 U (100 \u0026mu;l)\u003C\/td\u003E\n\u003Ctd class=\u0022product-price\u0022 data-product=\u0022E091\u0022 data-type=\u0022molecular\u0022\u003E$99.00\u003C\/td\u003E\n\u003C\/tr\u003E\n\u003Ctr\u003E\n\u003Ctd style=\u0022text-align: left;\u0022\u003E\u003Ca class=\u0022orange-link\u0022 href=\u0022\/poly-a-polymerase-yeast-e017.html\u0022 rel=\u0022noopener\u0022\u003EPoly(A) Polymerase, Yeast\u003C\/a\u003E\u003C\/td\u003E\n\u003Ctd style=\u0022text-align: left;\u0022\u003E\u003Ca class=\u0022orange-link\u0022 href=\u0022\/poly-a-polymerase-yeast-e017.html\u0022 rel=\u0022noopener\u0022\u003EE017\u003C\/a\u003E\u003C\/td\u003E\n\u003Ctd style=\u0022text-align: left;\u0022\u003E100 U (100 \u0026mu;l)\u003C\/td\u003E\n\u003Ctd class=\u0022product-price\u0022 data-product=\u0022E017\u0022 data-type=\u0022molecular\u0022\u003E$52.50\u003C\/td\u003E\n\u003C\/tr\u003E\n\u003Ctr\u003E\n\u003Ctd style=\u0022text-align: left;\u0022\u003E\u003Ca class=\u0022orange-link\u0022 href=\u0022\/polyA-polymerase-e.coli-e099.html\u0022 rel=\u0022noopener\u0022\u003EPoly(A) Polymerase, \u003Cem\u003EE. coli\u003C\/em\u003E\u003C\/a\u003E\u003C\/td\u003E\n\u003Ctd style=\u0022text-align: left;\u0022\u003E\u003Ca class=\u0022orange-link\u0022 href=\u0022\/polyA-polymerase-e.coli-e099.html\u0022 rel=\u0022noopener\u0022\u003EE099\u003C\/a\u003E\u003C\/td\u003E\n\u003Ctd style=\u0022text-align: left;\u0022\u003E25 U (25 \u0026micro;l)\u003C\/td\u003E\n\u003Ctd class=\u0022product-price\u0022 data-product=\u0022E099\u0022 data-type=\u0022molecular\u0022\u003E$125.00\u003C\/td\u003E\n\u003C\/tr\u003E\n\u003Ctr\u003E\n\u003Ctd style=\u0022text-align: left;\u0022\u003E\u003Ca class=\u0022orange-link\u0022 href=\u0022\/t7-rna-polymerase-e041.html\u0022 rel=\u0022noopener\u0022\u003ET7 RNA Polymerase\u003C\/a\u003E\u003C\/td\u003E\n\u003Ctd style=\u0022text-align: left;\u0022\u003E\u003Ca class=\u0022orange-link\u0022 href=\u0022\/t7-rna-polymerase-e041.html\u0022 rel=\u0022noopener\u0022\u003EE041\u003C\/a\u003E\u003C\/td\u003E\n\u003Ctd style=\u0022text-align: left;\u0022\u003E100 ul (5000 U)\u003C\/td\u003E\n\u003Ctd class=\u0022product-price\u0022 data-product=\u0022E041\u0022 data-type=\u0022molecular\u0022\u003E$68.00\u003C\/td\u003E\n\u003C\/tr\u003E\n\u003Ctr\u003E\n\u003Ctd style=\u0022text-align: left;\u0022\u003E\u003Ca class=\u0022orange-link\u0022 href=\u0022\/t4-gene-32-protein-e040-vin.html\u0022 rel=\u0022noopener\u0022\u003ET4 Gene 32 Protein\u003C\/a\u003E\u003C\/td\u003E\n\u003Ctd style=\u0022text-align: left;\u0022\u003E\u003Ca class=\u0022orange-link\u0022 href=\u0022\/t4-gene-32-protein-e040-vin.html\u0022 rel=\u0022noopener\u0022\u003EE040\u003C\/a\u003E\u003C\/td\u003E\n\u003Ctd style=\u0022text-align: left;\u0022\u003E100 \u0026mu;g\u003C\/td\u003E\n\u003Ctd class=\u0022product-price\u0022 data-product=\u0022E040\u0022 data-type=\u0022molecular\u0022\u003E$52.50\u003C\/td\u003E\n\u003C\/tr\u003E\n\u003Ctr\u003E\n\u003Ctd style=\u0022text-align: left;\u0022\u003E\u003Ca class=\u0022orange-link\u0022 href=\u0022\/thermolabile-udg.html\u0022 rel=\u0022noopener\u0022\u003EThermolabile UDG\u003C\/a\u003E\u003C\/td\u003E\n\u003Ctd style=\u0022text-align: left;\u0022\u003E\u003Ca class=\u0022orange-link\u0022 href=\u0022\/thermolabile-udg.html\u0022 rel=\u0022noopener\u0022\u003EE095\u003C\/a\u003E\u003C\/td\u003E\n\u003Ctd style=\u0022text-align: left;\u0022\u003E200 reactions\u003C\/td\u003E\n\u003Ctd class=\u0022product-price\u0022 data-product=\u0022E095\u0022 data-type=\u0022molecular\u0022\u003E$85.00\u003C\/td\u003E\n\u003C\/tr\u003E\n\u003Ctr\u003E\n\u003Ctd style=\u0022text-align: left;\u0022\u003E\u003Ca class=\u0022orange-link\u0022 href=\u0022\/gfp-purified-protein-000033p.html\u0022 rel=\u0022noopener\u0022\u003EGFP Purified Protein\u003C\/a\u003E\u003C\/td\u003E\n\u003Ctd style=\u0022text-align: left;\u0022\u003E\u003Ca class=\u0022orange-link\u0022 href=\u0022\/gfp-purified-protein-000033p.html\u0022 rel=\u0022noopener\u0022\u003E000033P\u003C\/a\u003E\u003C\/td\u003E\n\u003Ctd style=\u0022text-align: left;\u0022\u003E50 \u0026mu;g\u003C\/td\u003E\n\u003Ctd class=\u0022product-price\u0022 data-product=\u0022000033P\u0022 data-type=\u0022molecular\u0022\u003E$158.00\u003C\/td\u003E\n\u003C\/tr\u003E\n\u003Ctr style=\u0022background-color: #f8f9fa;\u0022 align=\u0022left\u0022 class=\u0022abm-page-menu only-hide\u0022 id=\u0022in-vitro-Kits-Proteases\u0022\u003E\n\u003Ctd colspan=\u00224\u0022 style=\u0022text-align: left;\u0022\u003E\u003Cspan style=\u0022color: #000;\u0022\u003E\u003Cstrong\u003EProteases\u003C\/strong\u003E\u003C\/span\u003E\u003C\/td\u003E\n\u003C\/tr\u003E\n\u003Ctr\u003E\n\u003Ctd style=\u0022text-align: left;\u0022\u003E\u003Ca class=\u0022orange-link\u0022 href=\u0022\/e027name.html\u0022 rel=\u0022noopener\u0022\u003ETEV Protease\u003C\/a\u003E\u003C\/td\u003E\n\u003Ctd style=\u0022text-align: left;\u0022\u003E\u003Ca class=\u0022orange-link\u0022 href=\u0022\/e027name.html\u0022 rel=\u0022noopener\u0022\u003EE027\u003C\/a\u003E\u003C\/td\u003E\n\u003Ctd style=\u0022text-align: left;\u0022\u003E100 \u0026mu;l\u003C\/td\u003E\n\u003Ctd class=\u0022product-price\u0022 data-product=\u0022E027\u0022 data-type=\u0022molecular\u0022\u003E$47.50\u003C\/td\u003E\n\u003C\/tr\u003E\n\u003Ctr\u003E\n\u003Ctd style=\u0022text-align: left;\u0022\u003E\u003Ca class=\u0022orange-link\u0022 href=\u0022\/enterokinase-cleavage-enzyme-mammalian-produced-g699.html\u0022 rel=\u0022noopener\u0022\u003EEnterokinase Cleavage Enzyme (Mammalian Produced)\u003C\/a\u003E\u003C\/td\u003E\n\u003Ctd style=\u0022text-align: left;\u0022\u003E\u003Ca class=\u0022orange-link\u0022 href=\u0022\/enterokinase-cleavage-enzyme-mammalian-produced-g699.html\u0022 rel=\u0022noopener\u0022\u003EG699\u003C\/a\u003E\u003C\/td\u003E\n\u003Ctd style=\u0022text-align: left;\u0022\u003E100 Units\u003C\/td\u003E\n\u003Ctd class=\u0022product-price\u0022 data-product=\u0022G699\u0022 data-type=\u0022molecular\u0022\u003E$115.00\u003C\/td\u003E\n\u003C\/tr\u003E\n\u003Ctr\u003E\n\u003Ctd style=\u0022text-align: left; width: 30.8635%;\u0022\u003E\u003Ca class=\u0022orange-link\u0022 href=\u0022\/proteinase-k-g029.html\u0022 rel=\u0022noopener\u0022\u003EProteinase K\u003C\/a\u003E\u003C\/td\u003E\n\u003Ctd style=\u0022text-align: left; width: 14.6197%;\u0022\u003E\u003Ca class=\u0022orange-link\u0022 href=\u0022\/proteinase-k-g029.html\u0022 rel=\u0022noopener\u0022\u003EG029\u003C\/a\u003E\u003C\/td\u003E\n\u003Ctd style=\u0022text-align: left; width: 27.1058%;\u0022\u003E1.0 ml; 20mg\/ml\u003C\/td\u003E\n\u003Ctd class=\u0022product-price\u0022 data-product=\u0022G029\u0022\u003E$92.00\u003C\/td\u003E\n\u003C\/tr\u003E\n\u003C\/tbody\u003E\n\u003C\/table\u003E\n\u003C\/div\u003E\n\u003C\/div\u003E\n\u003C\/div\u003E\n\u003C\/div\u003E","meta_title":"Molecular Biology Enzymes and Kits","meta_keywords":"Molecular Biology Enzymes and Kits","meta_description":"Molecular Biology Enzymes and Kits","deleted_at":null,"enable":"Y","parent_list":"27","table_name":null,"image":null,"independentPage":0,"top_type":1,"sort_order":null,"in_footer":1,"fid":null,"created_at":null,"updated_at":"2025-04-02 01:04:45"},"info":{"id":16615,"cat_no_base":"E027","parent_id":755,"description":"\u003Cp\u003E\u003Cstrong\u003Eabm\u0026rsquo;s TEV Protease\u003C\/strong\u003E is an enhanced version of the site-specific protease derived from \u003Cem\u003ETobacco Etch Virus\u003C\/em\u003E (TEV). This optimized enzyme offers superior \u003Cstrong\u003Eactivity\u003C\/strong\u003E, \u003Cstrong\u003Estability\u003C\/strong\u003E, and \u003Cstrong\u003Esite-specificity\u003C\/strong\u003E compared to the native form. TEV Protease cleaves fusion proteins with high precision at the \u003Cstrong\u003EGln-Gly\u003C\/strong\u003E or \u003Cstrong\u003EGln-Ser\u003C\/strong\u003E bond within the seven-amino acid recognition sequence \u003Cstrong\u003EGlu-Asn-Leu-Tyr-Phe-Gln-Gly\/Ser\u003C\/strong\u003E (ENLYFQ(G\/S)).\u003C\/p\u003E\n\u003Cp\u003EThe enzyme is highly effective across a broad range of temperatures (\u003Cstrong\u003E4\u0026ndash;30\u0026deg;C\u003C\/strong\u003E, with an optimum at \u003Cstrong\u003E30\u0026deg;C\u003C\/strong\u003E) and pH values (\u003Cstrong\u003E5.5\u0026ndash;9.0\u003C\/strong\u003E), enabling flexible experimental conditions. Under optimal conditions, up to \u003Cstrong\u003E99% cleavage\u003C\/strong\u003E can be achieved in just \u003Cstrong\u003E1\u0026ndash;2 hours\u003C\/strong\u003E. Additionally, the inclusion of a \u003Cstrong\u003E6X-His tag\u003C\/strong\u003E at the N-terminus allows for convenient removal of TEV Protease post-cleavage using \u003Cstrong\u003ENi-IDA Agarose Beads\u003C\/strong\u003E (\u003Ca href=\u0022\/ni-ida-agarose-resins-g250.html\u0022\u003ECat. No. G250\u003C\/a\u003E) via affinity chromatography.\u003C\/p\u003E\n\u003Ctable class=\u0022bootstrap-table bootstrap-table-hover abm-service-table abm-perfect-table2\u0022 style=\u0022margin: 30px 0;\u0022\u003E\n\u003Cthead\u003E\n\u003Ctr\u003E\n\u003Cth\u003E\u003Cstrong\u003EProduct Component\u003C\/strong\u003E\u003C\/th\u003E\n\u003Cth\u003E\u003Cstrong\u003EQuantity\u003C\/strong\u003E\u003C\/th\u003E\n\u003C\/tr\u003E\n\u003C\/thead\u003E\n\u003Ctfoot\u003E\n\u003Ctr\u003E\n\u003Ctd\u003ETEV Protease (10 U\/\u0026micro;l)\u003C\/td\u003E\n\u003Ctd\u003E100 \u0026micro;l\u003C\/td\u003E\n\u003C\/tr\u003E\n\u003Ctr\u003E\n\u003Ctd\u003E20X TEV Protease Reaction Buffer\u003C\/td\u003E\n\u003Ctd\u003E1.0 ml\u003C\/td\u003E\n\u003C\/tr\u003E\n\u003Ctr\u003E\n\u003Ctd\u003E100 mM DTT\u003C\/td\u003E\n\u003Ctd\u003E500 \u0026micro;l\u003C\/td\u003E\n\u003C\/tr\u003E\n\u003C\/tfoot\u003E\n\u003C\/table\u003E","disclaimer":null,"application":"\u003Cul\u003E\n\u003Cli\u003ECleavage of tags from recombinant fusion proteins containing a TEV recognition site\u003C\/li\u003E\n\u003Cli\u003EOne step affinity removal of His-tagged TEV after cleavage\u003C\/li\u003E\n\u003C\/ul\u003E","components":null,"cas9_origin":null,"concentration":"10U\/ul","enzymes_size":null,"genecraft_series":null,"guarantee":null,"population":null,"qc":null,"format_general":null,"including_screening_kit":null,"expression_system_general":null,"purity":null,"image":"\/t\/e\/tev-protease-corrected.png","insert_size":null,"shipping_conditions":null,"source_catalog_number":null,"inactivation_protocol":null,"led_viewer_compatibility":null,"unit_definition":null,"vector":null,"reaction_buffer":null,"storage_buffer":null,"caution":null,"storage_condition":null,"product_volume":null,"reporter":null,"safeview_series":null,"source_catno":"abm","stain_color":null,"supplier":"ABM","internal_supplier":null,"internal_note":null,"inventory_location":"Extra Freezer 1","note":null,"recommend":null,"depositor":null,"licensor_name":null,"licensor_contact_information":null,"contract_termination_date":null,"royalty_rates":null,"cas_type":null,"cas_origin":null,"cas_protein_marker":null,"source":null,"endotoxin_level":null,"additional_information":null,"titer":null,"nucleotide_format":null,"protocol_overview":null,"source_price":null,"created_at":"2022-09-09 07:12:40","updated_at":"2025-03-13 20:30:11","short_description":null,"reaction_definition":null,"specificity":null,"promotions":null},"maps":[],"media":[{"id":379474,"parent_id":755,"parent_type":"App\\Models\\CatalogBaseMolecular","file_path":"\/t\/e\/tev-protease-corrected.png","title":null,"text":null,"file_type":"image","alt":null,"url":null,"position":1,"status":0,"entity_id_m2":18084831,"sku_in_m2":"E027","value_id_m2":49910076,"attribute_id":90,"created_at":"2022-07-19 04:36:22","updated_at":"2023-04-10 05:39:28"}],"gene":null}