
Rapid Detection with Confidence!
Our new Mycoplasma Pro Detection Kit (Cat. No. G239) has a fast and simplified workflow, detects a market leading breadth of species, and has an internal PCR control built in to give you additional confidence in your results.
Competitor products not only entail a lengthier assembly process but also demand extended PCR run times, contributing to a less time-efficient workflow. Additionally, their results pose a challenge in terms of interpretation, introducing complexity to the analysis phase.
Market Leading Performance
| abm | Competitor A | Competitor S | |
|---|---|---|---|
| Sample type and Amount | Cell Supernatant, 2.5 ul | Cell suspension, 1 ml | Cell Supernatant, 100 ul |
| Number of reactions per kit | 100 | 40 | 24 |
| Sample Preparation Time | ~10 min | ~ 1 hr | ~ 20 min |
| PCR Run Time | 1 hr 10 min | 1 hr 37 min | 2 hr 11 min |
| Total Time to Results | 1 hr 20 min | 2 hr 37 min | 2 hr 31 min |
| Internal PCR control | Yes | No | Yes |
| Number of species | 300 + | 60+ | Not specified |
| Reported Sensitivity | 10 CFU/ml | 2 fg at 32 cycles | ≤20 mycoplasma genomes per 2 mL of sample |
| Ease of Result Readout | Straightforward | Challenging due to overlap of positive control and mycoplasma positive result in a sample | Challenging because internal PCR control decreases in brightness with increasing contamination level |
| Additional Materials Required | - | Heat block, Centrifuge | Taq DNA Polymerase, Heat Block, Centrifuge |
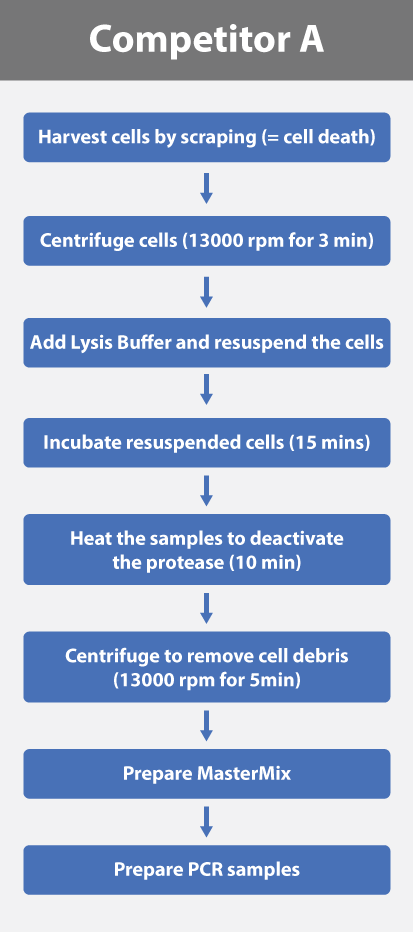
Streamlined and Simple Workflow


Trustworthy Screening Result
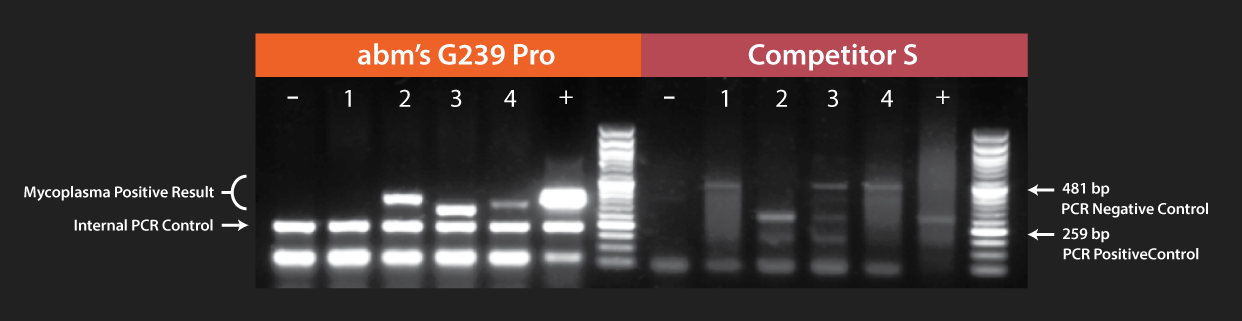
The inclusion of an internal PCR control is imperative for discerning true negative results from reactions affected by factors such as media additives, amplification conditions, or human error. Our Mycoplasma Pro Detection Kit (Cat. No. G239) addresses this necessity by incorporating an internal PCR control (180 bp band) that effectively showcases the functionality of the reaction. Notably, this internal PCR control is easily distinguishable from the Mycoplasma-positive PCR result, which falls within the range of 300-600 bp.
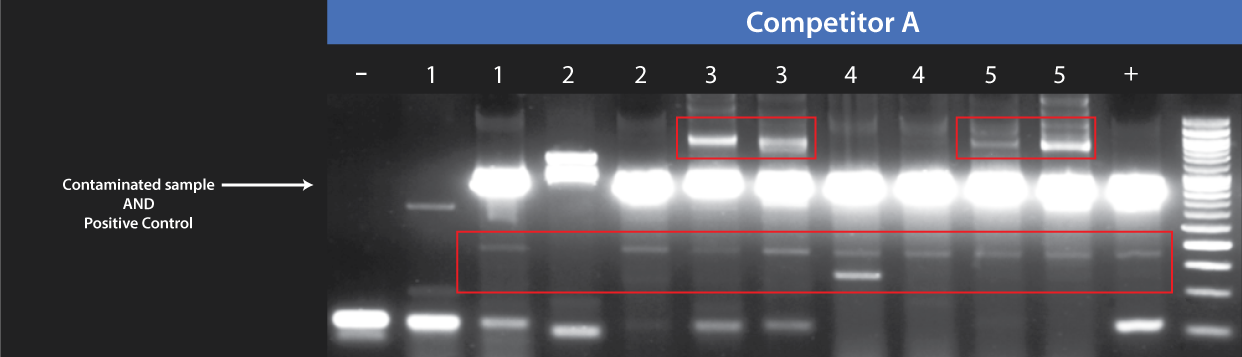
In contrast, Competitor A, whom we evaluated against our product, lacks an internal PCR control in their offering. Instead, they propose a separate PCR reaction for each sample with the addition of a positive control, specifically, “test sample + positive control.” However, this approach comes with two significant drawbacks. Firstly, it necessitates doubling the number of required assays – reaction 1 for the "test sample" and reaction 2 for the "test sample + positive control," leading to increased consumption of the provided positive control. Secondly, a notable concern arises due to the observed overlap of bands: their positive control is 464 bp, while the positive PCR result falls within the range of 434-468 bp. This overlapping scenario introduces challenges in the interpretation of results.
Definitive Result Read Out
Fast decision-making in cell culture management demands reliable results. The Mycoplasma Detection Kit from Competitor A introduces several challenges, including issues like excessive non-specific annealing and overlapping bands. Competitor S faces similar problems, with inconsistent intensity in the negative control as contamination levels rise. In contrast, abm's Mycoplasma Pro Detection Kit (Cat. No. G239) excels in sensitivity and specificity, providing users with unparalleled confidence in their results. The kit not only eliminates the drawbacks found in competing products but also ensures consistent and dependable outcomes.
Superior Performance at Best Value
abm's sample preparation is not only fast but also highly efficient. Researchers can expedite the process by simply mixing the kit components according to the desired number of assays and then adding the supernatant from the suspected cells. In contrast, competitors' products often involve cell lysis and/or denaturation, increasing both sample preparation time and the risk of contamination through the handling of potentially compromised cells.
Furthermore, our Mycoplasma Pro Detection Kit (Cat. No. G239) offers exceptional value, providing sufficient material for 100 reactions. The product comes complete with Blastaq™ 2X PCR MasterMix (Cat. No. G895), eliminating the need for additional reagents or equipment. In comparison, competitors impose limits of 24 and 40 assays, partly due to additional kit components such as lysis tubes and lyophilized mixtures. Competitor S also requires customers to separately purchase a Taq DNA Polymerase or use own polymerase. This not only results in higher pricing per reaction but also leaves less room for troubleshooting challenging samples. Our comprehensive solution not only streamlines the process but also ensures cost-effectiveness and reliability in Mycoplasma detection.
Order Information
| Product Name | Cat. No. | Size |
|---|---|---|
| Mycoplasma PCR Detection | ||
| Mycoplasma PCR Detection Kit | G238 | 100 rxn |
| Mycoplasma Pro PCR Detection Kit | G239 | 100 rxn |
| Mycoplasma Elimination | ||
| MycoAway™ Mycoplasma Elimination Cocktail (1000X) | G398 | 1.0 ml |
| Bundles | ||
| Mycoplasma PCR Detection & Elimination Bundle | G238-G398 | Bundle |
| Mycoplasma Pro PCR Detection & Elimination Bundle | G239-G398 | Bundle |
